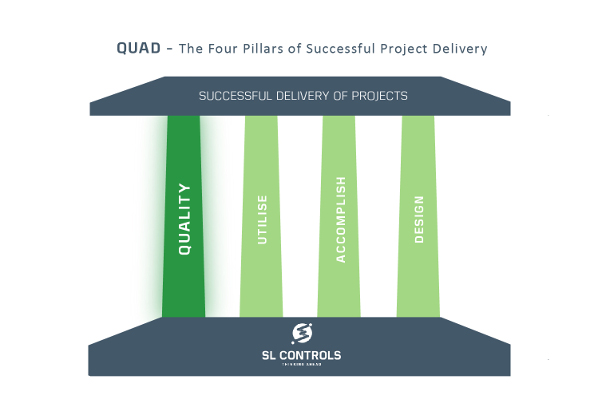
In the previous blog in this series, we outlined the four pillars of successful project delivery – QUAD. These pillars ensure major software projects, such as system automation and system control projects, meet expectations, complete on schedule and stay within budget. To recap, the four pillars are:
- Quality – the assurance of a successful project
- Utilise – enabling a successful project
- Accomplish – executing a successful project
- Design – the pre-requisite of successful projects
This blog focuses on one of those pillars: Quality, the assurance of a successful project.
Explaining the Quality Pillar
Quality strategies and actions taken throughout the entire project are crucial to its success. This begins at the requirements stage and continues through design, coding, testing, implementation and maintenance.
Also, at SL Controls, quality strategies and actions mean delivering projects in accordance with our ISO accredited QMS system.
From a client perspective, this results in expectations being met including functionality, performance, budget, and timeframe expectations.
Practical Steps
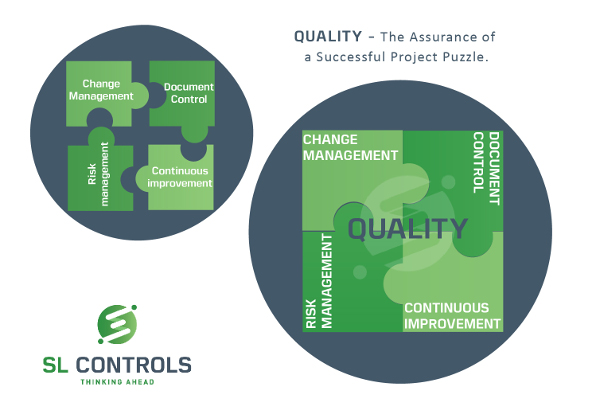
The practical quality steps required to deliver a successful project can be split into four broad areas:
- Change management
- Risk management
- Document control
- Continuous improvement
Let’s look at each element in more detail.
Change Management
Every software project you initiate will involve change control and management. As a result, change control and management should be an umbrella over every project.
Change management begins by identifying the impact on controlled items such as documentation, application software, operating software, firmware, hardware and configuration data associated with the project. This is one of the first things that should be done. Once you identify the impact to the controlled items, you can then make decisions on what you need to do from a quality perspective. This can include risk assessments, design, testing, deciding on the suite of documentation you will require, etc.
The change control process then tracks all this through to the completion of the project – right through to sign-off of all elements.
Risk Management
All Automation systems and control projects should take a risk-based approach. In fact, all software projects should have a risk-based approach. This means carrying out risk assessments on any and all aspects of the project.
Risk management on a project begins with an initial risk assessment. The purpose of this risk assessment is to determine the overall impact that the software may have on patient safety, product quality and data integrity. This risk assessment is typically followed by a functional risk assessment which involves calling out each function and then assessing the risk. It also includes safety risk assessments which involve employee, contractor, and, often, patient/customer safety. It is also important to do project and logistical risk assessments looking at time overruns, budget overruns, and other operational issues.
With each risk assessment, it is important to clearly identify the risk, understand how to mitigate the risk, and decide on the level of testing required.
Document Control
This part of the quality process covers document control and source code control, particularly version control of all code. For example, taking the existing code from a machine and then capturing, documenting, and controlling all updates to that code throughout the project. The same applies to other documents created during the project including design documents, test plans and more.
Good document control speeds up the error correction process and ensures full traceability. Full version management should continue until live implementation of the code and completion of the project.
Continuous Improvement
This is a central part of the SL Controls’ ethos and is crucial to successful project delivery. It is also part of our QMS systems. Improvements to our QMS are typically driven through outputs from other critical internal processes such as customer complaints, internal and external audits, customer feedback and non-conformances.
Typically, improvements are handled within the critical processes where they are raised, however, improvements that require medium to longer term fixes are generally transferred to our CAPA process. Under the CAPA process, a full investigation of the issue is carried out before identifying and implementing a long-term corrective action to ensure the same issue doesn’t happen again on this or any future project.
Benefits of Focusing on Quality
There are several benefits to adopting an approach that puts quality at the core of successful project delivery, rather than considering it as an add-on:
- Delivers expectations – the project will be completed successfully and will meet your objectives.
- Ensures constant improvement – the formal process of continuous improvement ensures you benefit from the collective learning of SL Controls’ engineers on multiple projects.
- On-time and on-budget – effective quality strategies and actions ensure delivery on these important goals.
- Saves you time – you will spend less management time dealing with issues in a project when it has a major focus on quality. In addition, you will avoid potential lost revenues through product recalls and associated reputational damage, equipment downtime, and more.
- Increased confidence in the solution – a focus on quality throughout the project ensures it runs smoothly and delivers what you expect, increasing your confidence in the solution.
The Q Pillar of the QUAD Structure
The QUAD approach to successful project delivery includes the principle that each of the four pillars is equally important. This demonstrates the importance of quality in all systems automation and control projects.
In upcoming articles, we will explore the remaining three pillars – utilise, accomplish, and design.





Comments are closed.